Pyridine and Methanol Can Act as Weak Bases
2.x: Organic Acids and Organic Bases
- Page ID
- 31392
Objective
After completing this section, you should be able to
- predict the relative acidity of two organic molecules from their structures.
- predict the relative basicity of ii organic molecules from their structures.
This page explains the acidity of simple organic acids and looks at the factors which touch their relative strengths.
Organic acids as weak acids
For the purposes of this topic, we are going to take the definition of an acid as "a substance which donates hydrogen ions (protons) to other things". We are going to get a measure of this past looking at how hands the acids release hydrogen ions to water molecules when they are in solution in water.
An acid in solution sets upwardly this equilibrium:
\[AH_{(aq)} + H_2O_{(fifty)} \rightleftharpoons A^-_{(aq)} + H_3O^+_{(aq)}\]
A hydronium ion is formed together with the anion (negative ion) from the acid. This equilibrium is sometimes simplified past leaving out the water to emphasize the ionization of the acid.
\[AH_{(aq)} \rightleftharpoons A^-_{(aq)} + H^+_{(aq)}\]
If you write it like this, you must include the country symbols - "(aq)". Writing H+ (aq) implies that the hydrogen ion is fastened to a water molecule equally H3O+. Hydrogen ions are ever fastened to something during chemical reactions.
The organic acids are weak in the sense that this ionization is very incomplete. At any one time, most of the acid volition be nowadays in the solution as un-ionized molecules. For example, in the case of dilute ethanoic acid, the solution contains well-nigh 99% of ethanoic acid molecules - at whatever instant, just about one% have really ionized. The position of equilibrium therefore lies well to the left.
Weak acrid polarization
Organic acids tin can usually can be characterized in electrostatic potential maps past the presence of of a positively polarized hydrogen atom shown in blue. When looking at the maps below, methanol has a slightly polarized O-H bond and is considered very weakly acidic. The O-H bond in methyl amine is less polarized, as shown past the lighter bluish color effectually the hydrogen, making it less acidic than methanol. However, the C-H bail in ethane lack near any polarity, as shown past the lack of a blue color, making it non-acidic. The following discussion will explain the difference in acerbity of these and other organic molecules molecules.
Comparison the strengths of weak acids
Acid strength is strongly correlated to stability of the cohabit base that volition form by removing a proton. In order to analyze how acidic a molecule is likely to exist, and so you need to estimate the stability of its cohabit base.
Stabilization of the Conjugate Base - Four Main Considerations:
- Size and electronegativity of the atom property the accuse
- Tin can the accuse be delocalized by resonance?
- Are there any inductive effects?
- Hybridization of orbital holding the charge
These considerations are listed in order of importance and are explained individually, but must be looked at collectively.
-
Size and Electronegativity Effects in Acidity
When comparing elements, it depends on the positional relationship of the elements on the periodic table. When moving a period (aka across a row) of the primary grouping elements, the valence electrons all occupy orbitals in the same shell. These electrons have comparable energy, so this factor does not aid united states of america discern differences relative stability. Differences in electronegativity are now the ascendant gene. This trend is shown when comparison the pKa values of ethane, methyl amine, and methanol which reflects the relative electronegativities of the C < N < O. The central to understanding this trend is to consider the hypothetical conjugate base in each case: the more than stable the conjugate base of operations, the stronger the acid. In general, the more than electronegative an atom, the better it is able to bear a negative charge. In the ethyl anion, the negative charge is borne past carbon, in the methylamine anion past nitrogen, and in the methoxide anion by an oxygen. Remember the periodic trend in electronegativity: it besides increases every bit nosotros move from left to right along a row, meaning that oxygen is the most electronegative of three elements being considered. This makes the negative charge on the methoxide anion the most stable of the 3 cohabit bases and methanol the strongest of the three acids. Besides, carbon is the least electronegative making ethane the weakest of the three acids.
Within a Group (aka down a column) As nosotros move downwardly the periodic table, the electrons are occupying higher energy subshells creating a larger atomic size and book. Equally the book of an chemical element increases, any negative accuse present tends to become more spread out which decreases electron density and increases stability. The figure below shows spheres representing the atoms of the south and p blocks from the periodic table to scale, showing the 2 trends for the atomic radius.
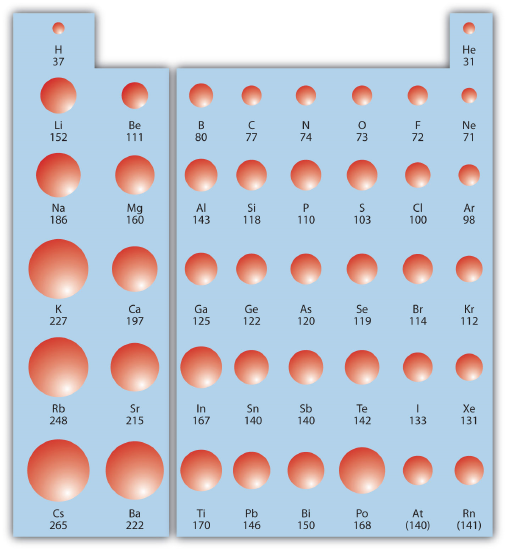
This relationship of atomic size and electron density is illustrated when nosotros compare the relative acidities of methanol, CH3OH, with methanethiol, CH3SH. The lower pKa value of 10.iv for methanethiol indicates that it is a stronger acrid than methanol with a pKa value of 15.5. It is important to remember that neither chemical compound is considered an acid. These relationships become useful when trying to deprotonate compounds to increase their chemic reactivity in not-aqueous reaction conditions.
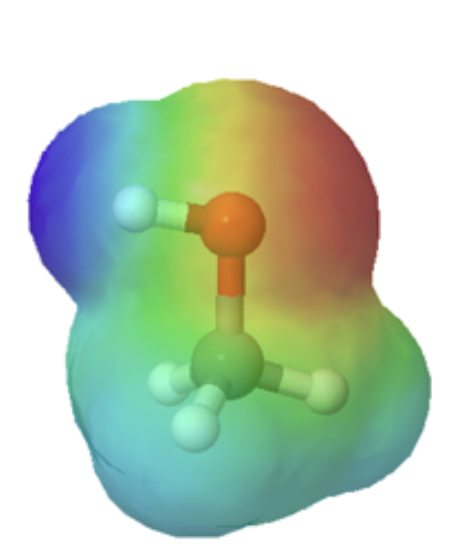
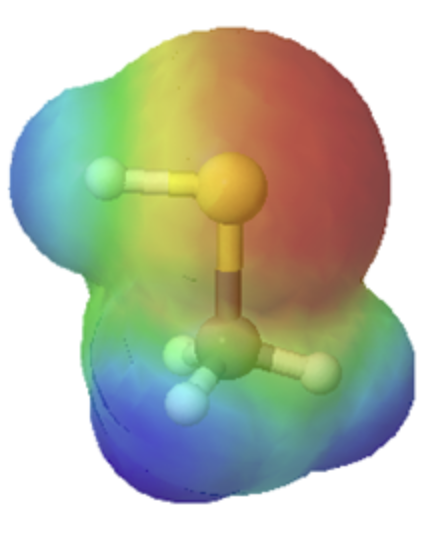
The departure in size can easily exist seen when looking at the electrostatic potential maps for methanol (Left) and methanethiol (Right). The sulfur atom methanethiol is larger than the oxygen atom in methanol. The larger size of sulfur will be improve able to delocalize and stabilize the negative accuse in its cohabit base of operations metanethiolate.
-
Resonance Effects in Acerbity
This section volition focus on how the resonance structures of different organic groups contributes to their relative acidity even though the same element acts equally the proton donor. When evaluating cohabit bases for the presence of resonance contributors, call back to expect for movable electrons (alone pairs and pi bonding electrons). Delocalizing electrons over two or more atoms spreads out the electron density, increasing the stability of the conjugate base, and increasing the acidity of the respective acid. A classic case compares the relative acidity of ethanol and acetic acid, only the conclusions we achieve will be equally valid for all alcohol and carboxylic acid groups. Despite the fact that they are both oxygen acids, the pKa values of ethanol and acetic acrid are very dissimilar.
In both species, the negative charge on the cohabit base is held past an oxygen, then periodic trends cannot be invoked. For acetic acid, all the same, there is a primal difference: a resonance correspondent can be drawn in which the negative charge is drawn on the second oxygen of the grouping. The two resonance forms for the conjugate base of operations are equal in energy, according to our 'rules of resonance' (Section 2.v). What this means is that the negative charge on the acetate ion is not located on one oxygen or the other: rather it is shared between the 2. Chemists utilize the term 'delocalization of charge' to describe this situation. In the ethoxide ion, by contrast, the negative charge is 'locked' on the single oxygen. This stabilization leads to a markedly increased acidity.
The delocalization of charge by resonance has a very powerful effect on the reactivity of organic molecules, plenty to business relationship for the departure of nearly 12 pKa units between ethanol and acerb acrid (and remember, pKa is a log expression, and so we are talking well-nigh a divergence of over 1012 betwixt the acidity constants for the 2 molecules). The acetate ion is much more stable than the ethoxide ion, due to the effects of resonance delocalization.
The effects of conjugation can be seen when comparison the electrostatic potential maps of ethanol and acetic acid. Conjugation creates a greater polarization in the O-H bond in acetic acrid as shown past its darker bluish color.
Why is Phenol Acidic?
Compounds like alcohols and phenol which contain an -OH grouping attached to a hydrocarbon are very weak acids. Alcohols are then weakly acidic that, for normal lab purposes, their acidity can exist about ignored. However, phenol is sufficiently acidic for it to have recognizably acidic properties - even if it is even so a very weak acid. A hydrogen ion can intermission away from the -OH group and transfer to a base of operations. For example, in aqueous solution:
Since phenol is a very weak acid, the position of equilibrium lies well to the left. However, phenol can lose a hydrogen ion because the phenoxide ion (or phenolate ion - the two terms tin be used interchangeably) formed is stabilized due to resonance. The negative accuse on the oxygen atom is delocalized around the ring since i of the lone pairs on the oxygen atom tin can exist in a p orbital and overlap with the pi electrons on the benzene band.
This overlap leads to a delocalization which extends from the ring out over the oxygen atom. As a result, the negative charge is no longer entirely localized on the oxygen, just is spread out around the whole ion. Spreading the charge around makes the ion more stable than information technology would be if all the accuse remained on the oxygen. However, oxygen is the most electronegative element in the ion and the delocalized electrons will be drawn towards information technology. That means that at that place will all the same be a lot of charge around the oxygen which volition tend to attract the hydrogen ion dorsum once more. That is why phenol is only a very weak acid.
This explains why phenol is a much stronger acid than cyclohexanol. Equally can be seen in the following energy diagram, resonance stabilization is increased for the conjugate base of phenol vs. cyclohexanol after removal of a proton.

The resonance stabilization in these two cases is very different. An important principle of resonance is that charge separation diminishes the importance of contributors to the resonance hybrid. The contributing structures to the phenol hybrid all suffer charge separation, resulting in very modest stabilization of this compound. On the other paw, the phenolate anion is already charged, and the approved contributors human activity to disperse the charge, resulting in a substantial stabilization of this species. The conjugate bases of uncomplicated alcohols are not stabilized past accuse delocalization, and then the acidity of these compounds is similar to that of water. An free energy diagram showing the effect of resonance on cyclohexanol and phenol acidities is shown on the right. Since the resonance stabilization of the phenolate conjugate base of operations is much greater than the stabilization of phenol itself, the acerbity of phenol relative to cyclohexanol is increased. Supporting show that the phenolate negative charge is delocalized on the ortho and para carbons of the benzene band comes from the influence of electron-withdrawing substituents at those sites.
Acidity of hydrogen α (alpha) to carbonyl
Alkyl hydrogen atoms bonded to a carbon atom in a α (alpha) position (directly adjacent) relative to a C=O group display unusual acerbity. While the pKa values for alkyl C-H bonds in is typically on the social club of xl-50, pKa values for these alpha hydrogens is more on the society of 19-20. This is almost exclusively due to the resonance stabilization of the product carbanion, called an enolate, every bit illustrated in the diagram beneath. The effect of the the stabilizing C=O is seen when comparing the pKa for the α hydrogens of aldehydes (~16-18), ketones (~19-21), and esters (~23-25).
-
Inductive Furnishings
The anterior issue is an experimentally observed effect of the transmission of charge through a chain of atoms in a molecule, resulting in a permanent dipole in a bond. For example, in a carboxylic acid group the presence of chlorine on side by side carbons increases the acidity of the carboxylic acrid group. A chlorine atom is more than electronegative than hydrogen, and thus is able to 'induce', or 'pull' electron density towards itself, abroad from the carboxylate group. This further spreads out the electron density of the cohabit base, which has a stabilizing result. In this context, the chlorine substituent is chosen an electron-withdrawing group. Detect that the pKa-lowering effect of each chlorine atom, while meaning, is not every bit dramatic as the delocalizing resonance effect illustrated by the departure in pKa values between an alcohol and a carboxylic acid. In general, resonance effects are more powerful than inductive effects .
negative charge is delocalized by being pulled out onto chlorine atom
Compare the pKa values of acetic acid and its mono-, di-, and tri-chlorinated derivatives:
The inductive effects of chlorine can be clearly seen when looking at the electrostatic potential maps of acetic acid (Left) and trichloroacetic acid (Right). The O-H bond in trichloroacetic acrid is highly polarized as shown by the nighttime blue color. This illustrates that tricholoracetic acid is a much stronger acid than acetic acrid.
Because the inductive event depends on electronegativity, fluorine substituents have a more pronounced pKa-lowering upshot than chlorine substituents.
In addition, the inductive takes place through covalent bonds, and its influence decreases markedly with distance – thus a chlorine 2 carbons away from a carboxylic acid group has a decreased effect compared to a chlorine only 1 carbon away. two-chloropropanoic acid has a pKa of 2.8 while for iii-chloropropanoic acid, the pKa is 4.0.
Alkyl groups (hydrocarbons) are weak inductive electron donators. In this instance the inductive effect pushes electron density onto the conjugate base of operations, causing the electron density to become more than concentrated and producing a destabilizing effect.
more than negative charge pushed towards already negative cease
The inductive effects of alkyl groups causes a significant variation in the acidities of different carboxylic acids. Notice that the inductive effect drops off afterwards the alkyl concatenation is about three carbons long.
| pKa | |
| HCOOH (Methanoic Acrid or Formic Acrid) | 3.75 |
| CHiiiCOOH (Ethanoic Acid or Acerb Acrid) | 4.76 |
| CH3CHiiCOOH (Propanoic Acrid) | 4.87 |
| CHiiiCHiiCH2COOH (Butanoic Acid) | four.82 |
-
Orbital Hybridization
The hybridization of an orbital affects its electronegativity. Within a trounce, the due south orbitals occupy the region closer to the nucleus than the p orbitals. Therefore, the spherical s orbitals are more electronegative than the lobed p orbitals. The relative electronegativity of hybridized orbitals is sp > spii > sp3 since the percentage of s-character is decreasing every bit more p-orbitals are added to the hybrids. This trend indicates the sp hybridized orbitals are more stable with a negative charge than sp3 hybridized orbitals. The table beneath shows how orbital hybridization influences relative acerbity.
| compound | conjugate base of operations | hybridization | s character | pKa | |
| | | spiii | 25% | 50 | weakest acid |
| | | sptwo | 33% | 44 | ↓ |
| | | 36 | ↓ | ||
| | | sp | fifty% | 25 | ↓ |
| | | 16 | strongest acid |
Comparison the Strengths of Weak Bases
Technically, organic bases are characterized by the presence of an cantlet with alone pair electrons. These lonely pairs contain a high electron density, which is shown carmine in the electrostatic potential maps, and can bond to H+. Below are the maps of methanol, methyl amine, and acetone. All three compounds tin be protonated with a sufficiently strong acid. Note, that all 3 of these compounds also accept the ability to donate a proton when reacted with a strong enough base. Whether these compounds human activity every bit a acid or base depends on the conditions.
It is mutual to compare basicity's quantitatively by using the pKa'south of their cohabit acids rather than their pKb'southward. Since pKa + pKb = 14, the higher the pKa the stronger the base, in contrast to the usual inverse relationship of pKa with acerbity. Call up that ammonia (NH3) acts as a base considering the nitrogen atom has a lone pair of electrons that can accept a proton. The conjugate acid of most simple alkyl amines accept pKa's in the range 9.5 to 11.0, and their water solutions are bones (accept a pH of 11 to 12, depending on concentration). This can be illustrated by the reaction below where an amine removes a proton from water to form substituted ammonium (e.thou. NH4 +) ions and hydroxide (OH−) ions:
Amines are i of the simply neutral functional groups which are considered basic. This is a direct consequence of the presence of the unshared electron pair on the nitrogen. The unshared electron pair is less tightly held by the nitrogen of an amine than the corresponding oxygen of an alcohol, which makes it more available to act equally a base. As a specific example, methylamine reacts with water to form the methylammonium ion and the OH− ion.
All of the have similarities to ammonia then nosotros'll get-go by looking at the reason for its basic properties. For the purposes of this topic, we are going to take the definition of a base of operations as "a substance which combines with hydrogen ions (protons)". We are going to get a measure out of this past looking at how easily the bases take hydrogen ions from h2o molecules when they are in solution in water.
Ammonia in solution sets up this equilibrium:
\[ NH_3 + H_2O \rightleftharpoons NH_4^+ + OH^-\tag{two.x.1}\]
An ammonium ion is formed together with hydroxide ions. Because the ammonia is only a weak base, it doesn't hang on to the extra hydrogen ion very effectively and and so the reaction is reversible. At whatsoever one time, about 99% of the ammonia is present as unreacted molecules. The position of equilibrium lies well to the left.
The ammonia reacts equally a base of operations because of the active lone pair on the nitrogen. Nitrogen is more electronegative than hydrogen then attracts the bonding electrons in the ammonia molecule towards itself. That ways that in addition to the solitary pair, there is a build-upward of negative charge around the nitrogen atom. That combination of extra negativity and active lone pair attracts the new hydrogen from the water.
When looking at the table below, it is clear that the basicity of nitrogen containing compounds is greatly influenced by their structures. The variance in the basicity of these compounds can mostly be explained past the effects of electron delocalization discussed above.
Tabular array \(\PageIndex{1}\): pKa of conjugate acids of a serial of amines.
| Compound | | | | | | | | | | |
|---|---|---|---|---|---|---|---|---|---|---|
| pKa | 11.0 | 10.7 | ten.7 | 9.iii | five.2 | four.vi | 1.0 | 0.0 | -1.0 | -10.0 |
Inductive Furnishings in Nitrogen Basicity
Alkylamines are more basic than ammonia since alkyl groups donate electrons to the more electronegative nitrogen. This anterior effect makes the electron density on the alkylamine nitrogen greater than the nitrogen of ammonium. That means that there will be a small amount of extra negative charge built up on the nitrogen atom. That extra negativity around the nitrogen makes the lone pair even more attractive towards hydrogen ions. Correspondingly, primary, secondary, and 3rd alkyl amines are more basic than ammonia.
methyl group pushes electron density toward the nitrogen, making it more basic
Making the nitrogen more than negative helps the solitary pair to choice up a hydrogen ion. What about the effect on the positive methylammonium ion formed? Is this more than stable than a elementary ammonium ion? Compare the methylammonium ion with an ammonium ion:
In the methylammonium ion, the positive accuse is spread around the ion by the "electron-pushing" issue of the methyl group. The more yous can spread accuse effectually, the more stable an ion becomes. In the ammonium ion at that place is non any style of spreading the accuse.
To summarize:
- The nitrogen is more negative in methylamine than in ammonia, and and then information technology picks up a hydrogen ion more readily.
- The ion formed from methylamine is more stable than the one formed from ammonia, and so is less likely to shed the hydrogen ion again.
Taken together, these hateful that methylamine is a stronger base than ammonia.
| Compound | pKa |
| NH3 | ix.3 |
| CH3NH2 | 10.66 |
| (CHthree)2NH | 10.74 |
| (CH3)3N | 9.81 |
Resonance Effects in Nitrogen Basicity
The resonance outcome also explains why a nitrogen atom is basic when it is in an amine, just not significantly basic when it is part of an amide group. While the lone pair of electrons in an amine nitrogen is localized in one place, the lone pair on an amide nitrogen is delocalized past resonance. The alone pair is stabilized by resonance delocalization. Here'due south another manner to think most information technology: the lone pair on an amide nitrogen is not bachelor for bonding with a proton – these two electrons are likewise stable being part of the delocalized pi-bonding system. The electrostatic potential map prove the effect of resonance on the basicity of an amide. The map shows that the electron density, shown in cerise, is nigh completely shifted towards the oxygen. This greatly decreases the basicity of the alone pair electrons on the nitrogen in an amide.
Aniline, the amine analog of phenol, is substantially less bones than an amine (every bit evidenced by the pKa of the conjugate acids).
| | aniline pKa of cohabit acid ~ v |
Nosotros can employ the aforementioned reasoning that we used when comparing the acidity of a phenol to that of an alcohol. In aniline, the lone pair on the nitrogen cantlet is stabilized by resonance with the aromatic pi organisation, making it less available for bonding and thus less basic.
lonely pair is stabilized through resonance
In these cases, you seem to be breaking the same oxygen-hydrogen bond each time, and so y'all might expect the strengths to be similar. The almost important factor in determining the relative acrid strengths of these molecules is the nature of the ions formed. You always get a hydronium ion - then that'due south constant - but the nature of the anion (the negative ion) varies markedly from example to instance.
Exercises
Do \(\PageIndex{i}\)
Select the more basic from each of the following pairs of compounds.
(a)
(b)
- Answer
-
a)
 The lone pair of electrons on the amide nitrogen are less bachelor to react with a proton.
The lone pair of electrons on the amide nitrogen are less bachelor to react with a proton.(b) NaOH -- The hydroxide has a negative accuse with 3 alone pairs of electrons that tin react with a proton.
Exercise \(\PageIndex{2}\)
The 4-methylbenzylammonium ion has a pKa of ix.51, and the butylammonium ion has a pKa of 10.59. Which is more basic? What's the pKb for each chemical compound?
- Answer
-
The butylammonium is more than basic. Remember that pKa+pKb = 14. The pKb for butylammonium is iii.41, the pKb for four-methylbenzylammonium is iv.49.
Source: https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Organic_Chemistry_%28McMurry%29/02:_Polar_Covalent_Bonds_Acids_and_Bases/2.10:_Organic_Acids_and_Organic_Bases

0 Response to "Pyridine and Methanol Can Act as Weak Bases"
Postar um comentário